The medical device industry is faced with multiple challenges – a shift in the industry from product-centric to patient-centric solutions, increasingly stringent industry regulations, declining margins, and competition from unconventional players – to name a few.
Additionally, digital technologies are assuming center stage in the functioning of medical devices, thereby influencing their development roadmaps. The blurring of boundaries between hardware, software, and network, as well as the interoperability requirements of connected ecosystems are leading to new challenges for enterprises in managing the development and maintenance of these devices.
To overcome these challenges, enterprises are increasingly looking toward service provider engagements. They expect service providers to not only provide the headcounts across hardware and software engineering, but to also offer domain expertise, best practices from other industries, reusable frameworks/accelerators, and lab infrastructure that enable a smooth and accelerated product development journey. The scope of engagements, which has traditionally been limited to device testing, regulatory compliance, and sustenance engineering, is now extending to service providers serving as extensions to enterprise R&D teams for prototypes, device connectivity, remote monitoring, and mobility enablement.
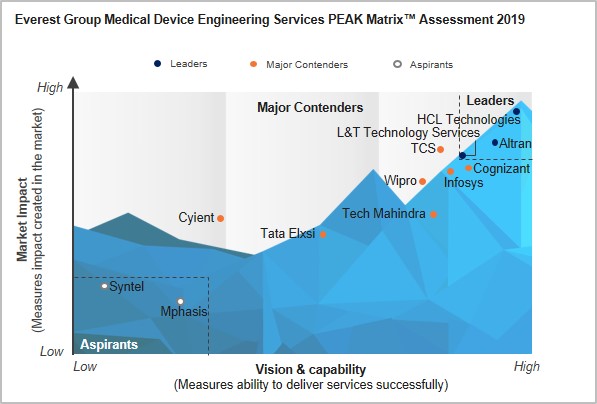
In this research, we present fact-based trends impacting the medical devices engineering services market, along with the assessment and detailed profiles of 12 leading service providers in this field, featured on the medical device engineering services PEAK Matrix.
Scope of this report
- Services: Medical device engineering services
- Geography: Global
- Service providers: 12 leading medical device engineering service providers
Contents
The research comprises three broad chapters:
- Medical device engineering services market trends:
- Medical device engineering services market size and projected growth
- Market breakdown by geographies, value chain elements, and service functions
- Analysis of key trends driving the growth in medical device engineering
- Key enterprise imperatives for engaging with service providers
- PEAK Matrix for medical device engineering services:
- Analysis of market impact and capabilities of 12 service providers based on the Everest Group PEAK MatrixTM, which is a composite index of a range of distinct metrics related to a service provider’s vision & capability and market impact
- Key characteristics of the three service provider categories–Leaders, Major Contenders, and Aspirants
- Insights into enterprise feedback for the three service provider categories
- Medical device engineering service provider profiles: Detailed profiles of 12 service providers that give a comprehensive picture of their medical devices engineering services vision, scale & scope of operations, key solutions, and partnerships
Membership(s)
Engineering Services
Life Sciences IT Services (ITS)